[Announcement] HGPI Endorsed “Joint Statement on the Implementation of Comprehensive Genomic Profiling test” (December 15, 2023)
date : 12/21/2023
Tags: NCDs, Precision Cancer Medicine
![[Announcement] HGPI Endorsed “Joint Statement on the Implementation of Comprehensive Genomic Profiling test” (December 15, 2023)](https://hgpi.org/en/wp-content/uploads/sites/2/ncd-pcm-20231215-top.png)
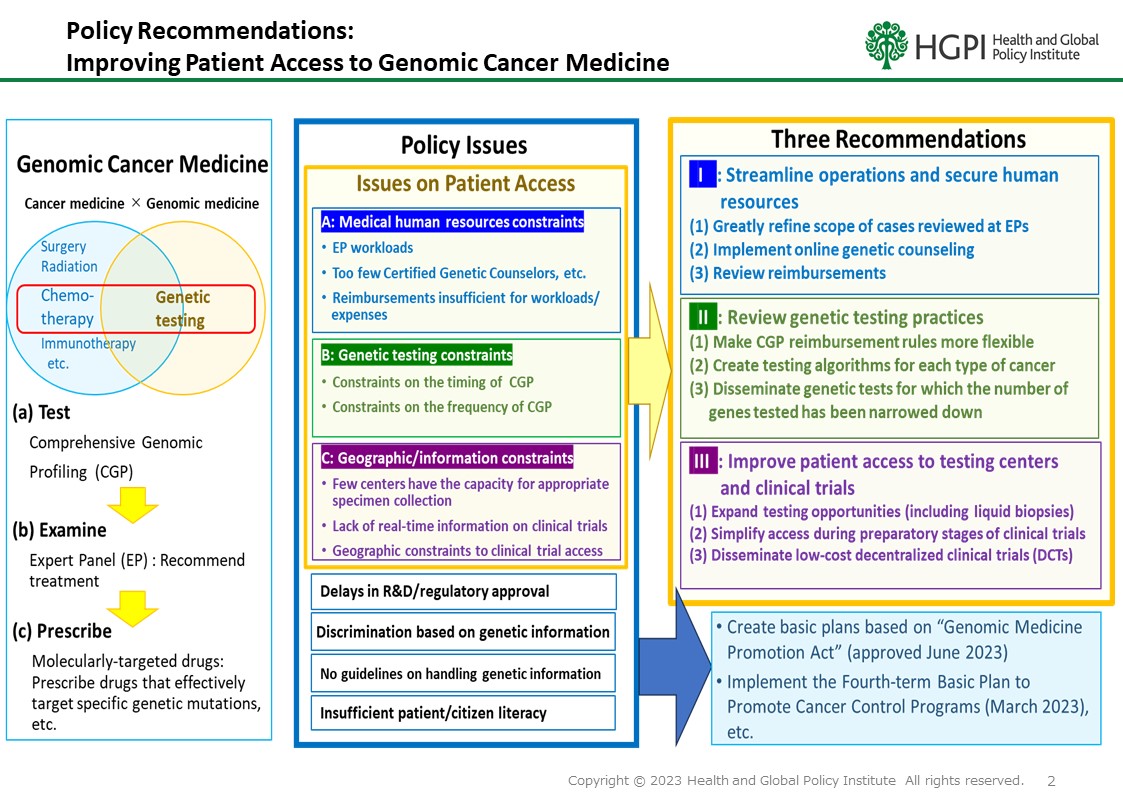
On December 15, 2023, Health and Global Policy Institute (HGPI) endorsed “Joint Statement on the Implementation of Comprehensive Genomic Profiling”.
In addition to our Organization, this Joint Statement is endorsed by the Japan Federation of Cancer Patient Groups, the European Federation of Pharmaceutical Industries and Associations (EFPIA Japan), the Pharmaceutical Research and Manufacturers of America (PhRMA), the Japanese Society of Medical Oncology, the Japanese Society of Clinical Oncology, the Japanese Cancer Association, and the Social Insurance Union of Societies Related to Internal Medicine.
This joint statement will be submitted to the Minister of Health, Labour and Welfare, Mr. Keizo Takemi, and other relevant officials.
Currently, the cancer gene panel tests (cancer Comprehensive Genomic Profiling tests) approved for use under the Japanese Health insurance system is only for ” solid tumors for which no standard treatment is available, or with local progression or metastasis for which standard treatment has been terminated (including those for whom termination is expected)”. Under these circumstances, there have been unfortunate cases in which patients have missed treatment opportunities due to deterioration of their general condition, etc., even when a cancer gene panel tests has revealed a gene mutation that can be treated with drug therapy, and it is too late to do so. In order to ensure that patients who need the most appropriate treatment can receive it at the most appropriate time, a number of proposals have been made by related organizations to resolve issues related to insurance reimbursement. We share a strong awareness of these issues and strongly request that the following points in particular be implemented in the FY2024 revision of the medical service fee.
Request
Expand insurance coverage for cancer gene panel testing so that it can be done at the appropriate time from the time of initial treatment.
Please note that the joint statement documents are only available in Japanese.
Top Research & Recommendations Posts
- [Research Report] Energy Conservation and Greenhouse Gas Emissions Reduction in Healthcare Facilities: Case Studies from New Construction and Facility Renewal (March 16, 2026)
- [Public Comment Submission] “The 7th Science, Technology and Innovation Basic Plan” (Draft) (February 19, 2026)
- [Research Report] Perceptions, Knowledge, Actions and Perspectives of Healthcare Organizations in Japan in Relation to Climate Change and Health: A Cross-Sectional Study (November 13, 2025)
- [Research Report] The 2025 Public Opinion Survey on Healthcare in Japan (March 17, 2025)
- [Policy Recommendations] Mental Health Project: Recommendations on Three Issues in the Area of Mental Health (July 4, 2025)
- [Research Report] The 2026 Public Opinion Survey on Healthcare in Japan (February 13, 2026)
- [Policy Recommendations] Dementia Project “The Future of Dementia Policy Surrounding Families and Others Who Care for People with Dementia” (April 27, 2026)
- [Policy Recommendations] Developing a National Health and Climate Strategy for Japan (June 26, 2024)
- [Policy Recommendations] Policy Recommendations in the Field of Blood Disorders “Building a Healthcare Ecosystem Centered on Patients and Those Affected” (April 13, 2026)
- [Research Report] The Public Opinion Survey on Child-Rearing in Modern Japan (Final Report) (March 4, 2022)
Featured Posts
-
2026-05-13
[Registration Open] HGPI Salon 2025-2026 Looking Ahead to the Future of Japan’s Social Security System “Session 5: The Value of Healthcare in Society – Perspectives from the Reiwa Era” (June 29, 2026)
![[Registration Open] HGPI Salon 2025-2026 Looking Ahead to the Future of Japan’s Social Security System “Session 5: The Value of Healthcare in Society – Perspectives from the Reiwa Era” (June 29, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/5thHGPISalon_20260629.png)
-
2026-05-15
[Registration Open] (Webinar) The 2nd J-PEP Seminar – A New Framework for Providing Information on Clinical Trial Participant Recruitment – Legal Points to Consider from the Perspective of Meaningful Involvement (June 15, 2026)
![[Registration Open] (Webinar) The 2nd J-PEP Seminar – A New Framework for Providing Information on Clinical Trial Participant Recruitment – Legal Points to Consider from the Perspective of Meaningful Involvement (June 15, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20260615_The2nd-J-PEPseminar.jpg)
-
2026-05-18
[Registration Open] Kick-off Panel Discussion “From Japan and Africa, in Discussion with the World: Cultivating Together a Philosophical Dialogue on Health” (June 15, 2026)
![[Registration Open] Kick-off Panel Discussion “From Japan and Africa, in Discussion with the World: Cultivating Together a Philosophical Dialogue on Health” (June 15, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20260615_Eyecathch_panel-discussion-From-Japan-and-Africa-in-Discussion-with-the-World‗02.png)
-
2026-05-19
[Registration Open] (Webinar) HGPI Special Seminar “Building a Society Where People Live with Blood Disorders: The Future of Blood Disorder Policy as Examined from the Frontlines of Home-Based Care × Lived Experience Research” (June 12, 2026)
![[Registration Open] (Webinar) HGPI Special Seminar “Building a Society Where People Live with Blood Disorders: The Future of Blood Disorder Policy as Examined from the Frontlines of Home-Based Care × Lived Experience Research” (June 12, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20260515_HGPI-Special-Seminar-Blood-Disorders.png)