[Policy Recommendations] Improving Patient Access to Genomic Cancer Medicine (August 10, 2023)
date : 8/10/2023
Tags: NCDs, Precision Cancer Medicine
![[Policy Recommendations] Improving Patient Access to Genomic Cancer Medicine (August 10, 2023)](https://hgpi.org/en/wp-content/uploads/sites/2/ncd-20230810-figure1_ENG.jpg)
Health and Global Policy Institute (HGPI) has presented a policy recommendation paper regarding the improvement of patient access to Genomic Cancer Medicine.
Executive summary
- Genomic Cancer Medicine was viewed as an advanced form of medical treatment when Comprehensive Genomic Profiling (CGP; or cancer gene panel tests) was granted health insurance coverage in 2019. Since then, people serving in clinical settings have accumulated experience handling this form of treatment. We are now at a stage to make Genomic Cancer Medicine a form of technology from which anyone should be able to benefit. To achieve this, we should improve patient access to deliver Genomic Cancer Medicine more broadly to the public.
- From the perspective of patient access opportunities, policy challenges with respect to delivering Genomic Cancer Medicine may be sorted into three categories:
A: Constraints related to human resources in medicine
B: Constraints related to genetic testing
C: Constraints related to geographic factors or information
We must take wherever possible actions to address each issue one by one, and expand opportunities for patients to access Genomic Cancer Medicine.
- Based on these understandings, HGPI offers the following recommendations:
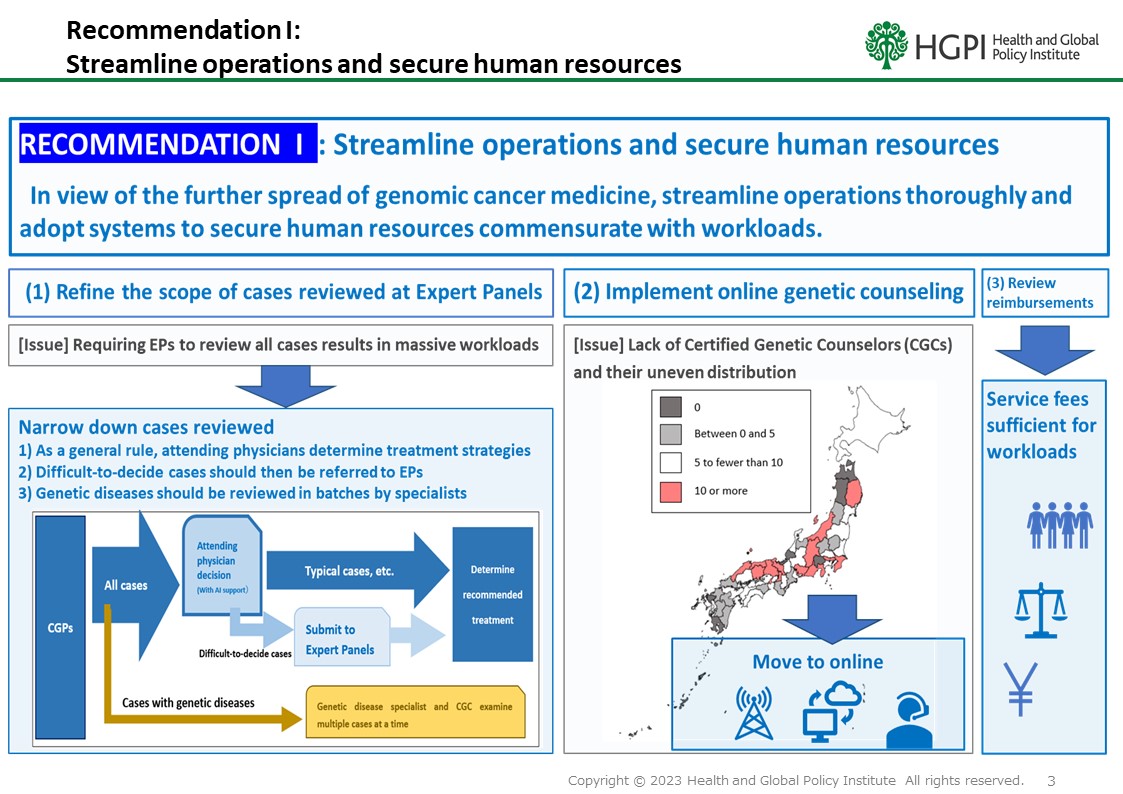
RECOMMENDATION I: Streamline operations and secure human resources
In view of the further spread of genomic cancer medicine, streamline operations thoroughly, and adopt systems to secure human resources commensurate with workloads.
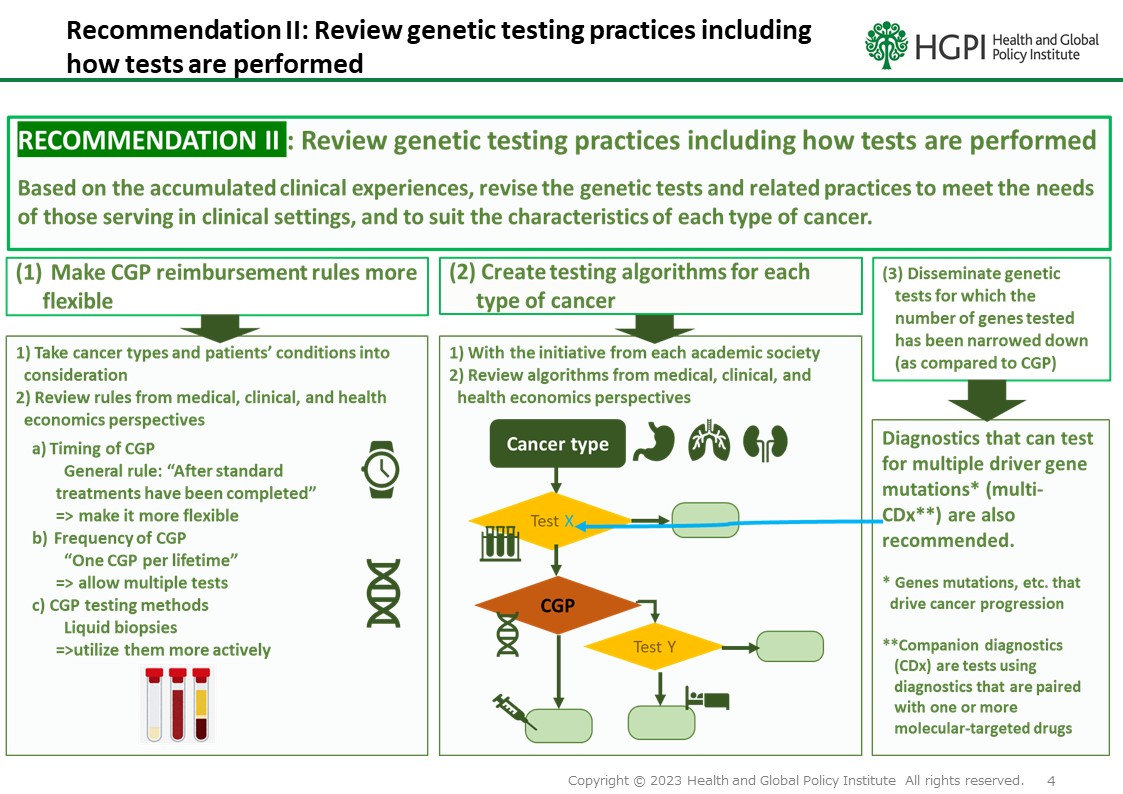
RECOMMENDATION II: Review genetic testing practices including how tests are performed
Based on the accumulated clinical experiences, revise the genetic tests and related practices to meet the needs of those serving in clinical settings, and to suit the characteristics of each type of cancer.
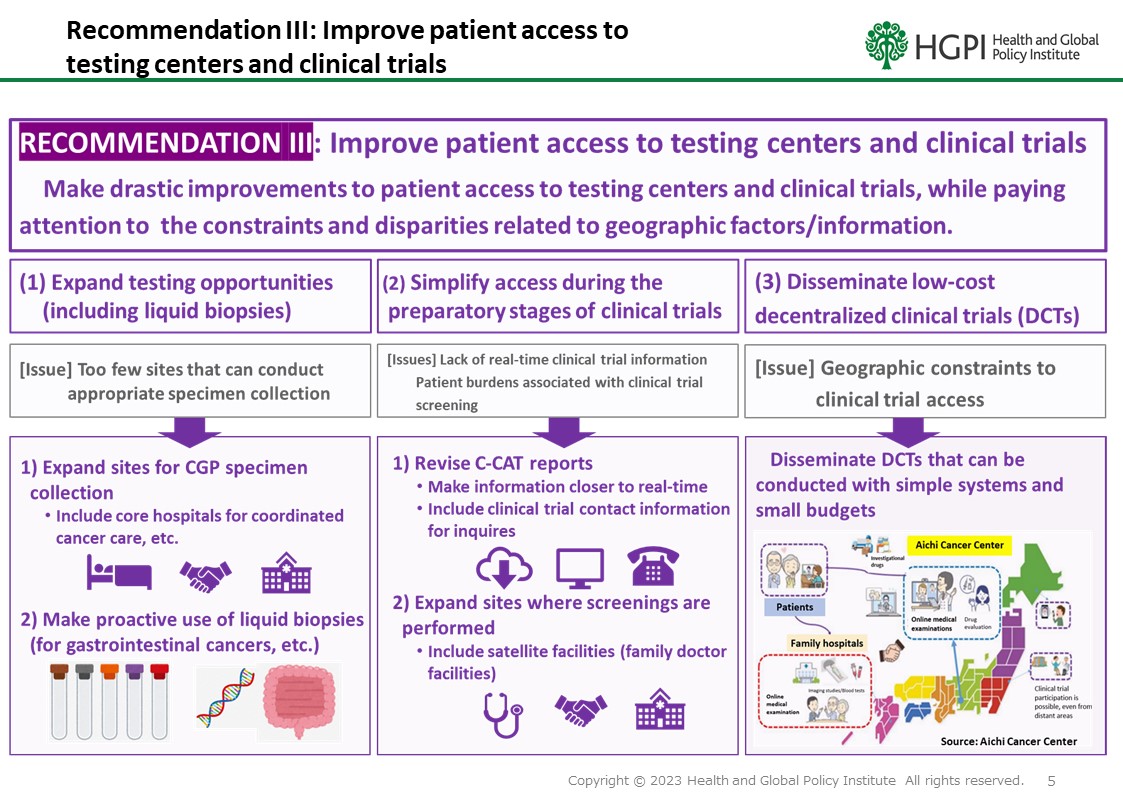
RECOMMENDATION III: Improve patient access to testing centers and clinical trials
While paying attention to the constraints and disparities related to geographic factors/information, make drastic improvements to patient access to testing centers and clinical trials.
As for precision cancer medicine, HGPI launched an initiative in FY2021 (“Project for Considering the Future of Precision Medicine with Industry, Government, Academia, and Civil Society”), and has made expert hearings and conducted surveys to examine the issue. Based on our findings, we offered comprehensive policy recommendations in September 2022.
For the second phase of this project, we compiled this policy recommendation paper, based on additional, expansive surveys and hearings with our focus on measures for better patient access to Genomic Cancer Medicine.
HGPI strongly hopes these recommendations will be utilized in developing further policy measures for Genomic Cancer Medicine, and patient-centered healthcare.
For details, please see the PDFs linked below.
Sponsors (in alphabetical order):
Bayer Yakuhin, Ltd.
Chugai Pharmaceutical Co., Ltd.
Janssen Pharmaceutical K.K.
MSD K.K.
Top Research & Recommendations Posts
- [Research Report] Energy Conservation and Greenhouse Gas Emissions Reduction in Healthcare Facilities: Case Studies from New Construction and Facility Renewal (March 16, 2026)
- [Public Comment Submission] “The 7th Science, Technology and Innovation Basic Plan” (Draft) (February 19, 2026)
- [Research Report] Perceptions, Knowledge, Actions and Perspectives of Healthcare Organizations in Japan in Relation to Climate Change and Health: A Cross-Sectional Study (November 13, 2025)
- [Research Report] The 2025 Public Opinion Survey on Healthcare in Japan (March 17, 2025)
- [Policy Recommendations] Mental Health Project: Recommendations on Three Issues in the Area of Mental Health (July 4, 2025)
- [Research Report] The 2026 Public Opinion Survey on Healthcare in Japan (February 13, 2026)
- [Policy Recommendations] Dementia Project “The Future of Dementia Policy Surrounding Families and Others Who Care for People with Dementia” (April 27, 2026)
- [Policy Recommendations] Developing a National Health and Climate Strategy for Japan (June 26, 2024)
- [Policy Recommendations] Policy Recommendations in the Field of Blood Disorders “Building a Healthcare Ecosystem Centered on Patients and Those Affected” (April 13, 2026)
- [Research Report] The Public Opinion Survey on Child-Rearing in Modern Japan (Final Report) (March 4, 2022)
Featured Posts
-
2026-05-13
[Registration Open] HGPI Salon 2025-2026 Looking Ahead to the Future of Japan’s Social Security System “Session 5: The Value of Healthcare in Society – Perspectives from the Reiwa Era” (June 29, 2026)
![[Registration Open] HGPI Salon 2025-2026 Looking Ahead to the Future of Japan’s Social Security System “Session 5: The Value of Healthcare in Society – Perspectives from the Reiwa Era” (June 29, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/5thHGPISalon_20260629.png)
-
2026-05-15
[Registration Open] (Webinar) The 2nd J-PEP Seminar – A New Framework for Providing Information on Clinical Trial Participant Recruitment – Legal Points to Consider from the Perspective of Meaningful Involvement (June 15, 2026)
![[Registration Open] (Webinar) The 2nd J-PEP Seminar – A New Framework for Providing Information on Clinical Trial Participant Recruitment – Legal Points to Consider from the Perspective of Meaningful Involvement (June 15, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20260615_The2nd-J-PEPseminar.jpg)
-
2026-05-18
[Registration Open] Kick-off Panel Discussion “From Japan and Africa, in Discussion with the World: Cultivating Together a Philosophical Dialogue on Health” (June 15, 2026)
![[Registration Open] Kick-off Panel Discussion “From Japan and Africa, in Discussion with the World: Cultivating Together a Philosophical Dialogue on Health” (June 15, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20260615_Eyecathch_panel-discussion-From-Japan-and-Africa-in-Discussion-with-the-World‗02.png)
-
2026-05-19
[Registration Open] (Webinar) HGPI Special Seminar “Building a Society Where People Live with Blood Disorders: The Future of Blood Disorder Policy as Examined from the Frontlines of Home-Based Care × Lived Experience Research” (June 12, 2026)
![[Registration Open] (Webinar) HGPI Special Seminar “Building a Society Where People Live with Blood Disorders: The Future of Blood Disorder Policy as Examined from the Frontlines of Home-Based Care × Lived Experience Research” (June 12, 2026)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20260515_HGPI-Special-Seminar-Blood-Disorders.png)