[HGPI Policy Column] No. 33 – From the NCDs (Precision Cancer Medicine) Team, What is precision cancer medicine?
date : 2/2/2023
![[HGPI Policy Column] No. 33 – From the NCDs (Precision Cancer Medicine) Team, What is precision cancer medicine?](https://hgpi.org/en/wp-content/uploads/sites/2/column-33-top.png)
1. In treating cancer, there are three major therapies, namely, (i) Surgical operation, (ii) Radiation therapy, and (iii) Drug therapy (Chemotherapy). Recently, treatment options have been increasing—for example, (iv) Cancer immunotherapy, which uses treatments to stimulate or restore the ability of the immune system to fight cancer, has become one of the major treatment options (see table below).
“Precision cancer medicine,” which has been attracting attention in recent years, is particularly related to (iii) Drug therapy. They identify the mutated genes, cancer cells of individual patients to be treated in advance, and then, “molecular-targeted drugs” (drugs that are particularly effective on those mutated genes and molecules) are selected and administered.
In conventional cancer drug therapy, cancers are usually perceived in terms of the affected organ, and drugs are administered with the expectation they will be effective for that type of cancer. In precision cancer medicine, they provide treatment in a more targeted manner, by focusing on the section of the gene where mutations are occurring. This is why it is called “precision” medicine.
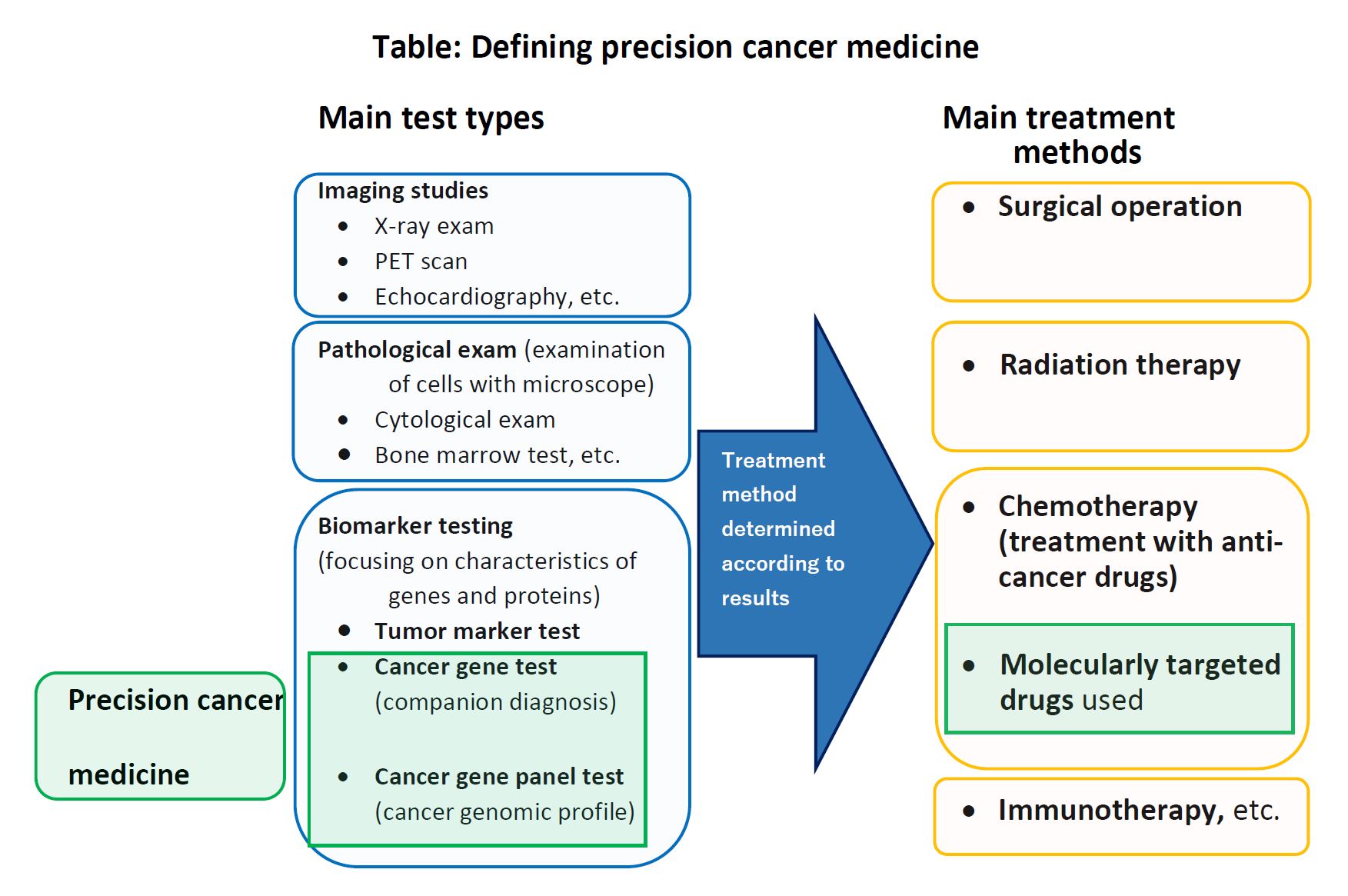
2. To carry out precision cancer medicine, genetic testing is indispensable. There are two methods of genetic testing for cancer: (i) “Cancer gene testing “or “Companion diagnostic testing” and (ii) “Cancer gene panel testing” or “Cancer genome profiling test.” Cancer gene testing determines the presence or absence of mutations in one or several genes, in order to determine in advance whether a certain therapeutic drug will be effective for a patient’s cancer. Cancer gene panel testing is to determine the presence or absence of mutations in a large number of genes (usually more than one hundred) at once.
3. Compared to conventional cancer drug therapy, more targeted therapies are administered in precision cancer medicine, and this can reduce burdens on patients, increase response rates, and achieve better outcomes. There are high expectations for the future development of precision cancer medicine.
On the other hand, there are many challenges: (a) the costs of testing and treatment are often quite expensive; (b) even if a genetic testing is conducted, it may not find a genetic mutation that is eligible for precision cancer medicine (in such a case, conventional drug therapy is an option); (c) nationwide, specialized facilities and human resources to conduct genetic testing and treatment are not that abundant; and (d) laws and regulations to strictly manage genetic information and to prevent genetic discrimination have not yet been fully established.
In order to further develop precision cancer medicine in the future, it is necessary to resolve these various issues as soon as possible.
Column authors
- Sayaka Honda (Program Specialist, HGPI)
- Osamu Takemoto (Program Specialist, HGPI)
- Haruka Sakamoto (Senior Manager, HGPI)
HGPI Policy Column (No.32) -From thePlanetary Health Policy Team- >
日本語
Top Research & Recommendations Posts
- [Announcement] A Turning Point Towards Building Green Healthcare Systems (June 5, 2024)
- [Policy Recommendations] Developing a National Health and Climate Strategy for Japan (June 26, 2024)
- [Publication Report] Guidance on Patient and Public Involvement (PPI) in Health Policymaking: Necessary Initiatives and Good Examples from the Public and Government (March 31, 2024)
- [Policy Recommendations] Obesity Control Promotion Project 2023 “The Next Steps for Engaging and Cooperating with Patients, Citizens, and Communities for Implements of Obesity Control Measurements” (April 8, 2024)
- [Research Report] Healthcare DX Project Research Report of Interviews ”Expectations for the Coming Era of Healthcare DX from People Living with Health Concerns” (June 10, 2024)
- [Policy Recommendations] Recommendation for the Basic Policy on Economic and Fiscal Management and Reform 2024 (June 11, 2024)
- [Policy Recommendations] Kidney Disease Control Promotion Project 2023 “Establishing Kidney Disease Control Measures with Patient, Citizen, and Community Engagement and Collaboration” Policy Recommendations, a Collection of Good Practices of Chronic Kidney Disease (CKD) and Control Measures in Local Governments (February 14, 2024)
- [Public Comment Submission] Web based consultations on NCDs and mental health by World Health Organization (June 25, 2024)
- [Research Report] Building a Mental Health Program for Children and Measuring its Effectiveness (June 16, 2022)
- [Research Report] The 2023 Public Opinion Survey on Satisfaction in Healthcare in Japan and Healthcare Applications of Generative AI (January 11, 2024)
Featured Posts
-
2024-06-21
[Event Report] HGPI Special Seminar – HGPI Celebrates its 20th Anniversary: Reflecting on HGPI’s Journey from the Past to the Future (January 16, 2024)
![[Event Report] HGPI Special Seminar – HGPI Celebrates its 20th Anniversary: Reflecting on HGPI’s Journey from the Past to the Future (January 16, 2024)](https://hgpi.org/en/wp-content/uploads/sites/2/240116_HGPISeminar_eyecatch-1.png)
-
2024-06-25
[Public Comment Submission] Web based consultations on NCDs and mental health by World Health Organization (June 25, 2024)
![[Public Comment Submission] Web based consultations on NCDs and mental health by World Health Organization (June 25, 2024)](https://hgpi.org/en/wp-content/uploads/sites/2/ncd-ppi-ph-mh-20240625-top.png)
-
2024-06-26
[Event Report] Global Expert Meeting “Innovation in Cancer Care in Developing Countries ~City Cancer Challenge Initiatives“ (June 20, 2024)
![[Event Report] Global Expert Meeting “Innovation in Cancer Care in Developing Countries ~City Cancer Challenge Initiatives“ (June 20, 2024)](https://hgpi.org/en/wp-content/uploads/sites/2/ncd-pcm-20240620-1-top-PXL_20240620_061249805.MP_-scaled-1.jpg)
-
2024-06-26
[Registration Open] (Webinar) The 127th HGPI Seminar: Current Issues and Future Prospects in Establishing a Health System and Protecting Public Health Through Policy (July 18, 2024)
![[Registration Open] (Webinar) The 127th HGPI Seminar: Current Issues and Future Prospects in Establishing a Health System and Protecting Public Health Through Policy (July 18, 2024)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20240718_127thHGPISeminar.png)
-
2024-07-01
[Registration Open] Meaningful Involvement Promotion Project Online Expert Meeting “Shaping the Future of Health Policy with People with Lived Experience and Citizens” (July 26, 2024)
![[Registration Open] Meaningful Involvement Promotion Project Online Expert Meeting “Shaping the Future of Health Policy with People with Lived Experience and Citizens” (July 26, 2024)](https://hgpi.org/en/wp-content/uploads/sites/2/HGPI_20240726_Shaping-the-Future-of-Health-Policy-with-People-with-Lived-Experience-and-Citizens.jpg)




